 
Here we further explore the nature of this state function and define it mathematically. In Chapter 13, we introduced the concept of entropy in relation to solution formation. To help explain why these phenomena proceed spontaneously in only one direction requires an additional state function called entropy (S), a thermodynamic property of all substances that is proportional to their degree of "disorder". 
Moreover, the molecules of a gas remain evenly distributed throughout the entire volume of a glass bulb and never spontaneously assemble in only one portion of the available volume. dQ dE + p dV where p is the pressure and V is the volume of the gas. Go, for the change using the following equation. 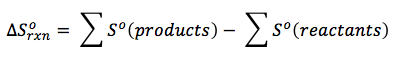
Substituting for the definition of work for a gas. From Free Energies of Formation From Enthalpy Changes and Entropy Changes From Equilibrium Constants. We begin by using the first law of thermodynamics: dE dQ - dW where E is the internal energy and W is the work done by the system. For example, after a cube of sugar has dissolved in a glass of water so that the sucrose molecules are uniformly dispersed in a dilute solution, they never spontaneously come back together in solution to form a sugar cube. For gases, there are two possible ways to evaluate the change in entropy. Also, in a constant volume process, TdS dU so that T ( S T) V ( U T) V C V. The temperatures associated with these states are T A T A and T B, T B, respectively. Imagine a system making a transition from state A to B in small, discrete steps. From a Maxwell relation (equation 12.6.15), ( S V) T ( P T) V. The change in entropy of a system for an arbitrary, reversible transition for which the temperature is not necessarily constant is defined by modifying S Q/T S Q / T. Therefore (13.4.2) T d S T ( S V) T d V + T ( S T) V d T. For a full video: see Thus enthalpy is not the only factor that determines whether a process is spontaneous. Let us first express entropy as a function of V and T (13.4.1) d S ( S V) T d V + ( S T) V d T. When water is placed on a block of wood under the flask, the highly endothermic reaction that takes place in the flask freezes water that has been placed under the beaker, so the flask becomes frozen to the wood. Solution Entropy change of an ideal gas From the Gibbsian equations, the change of entropy of an ideal gas can be expressed as Cases with constant specific heats When specific heats are constant, the integration can be simplified: Example Air is compressed from an initial state of 100 kPa and 300 K to 500 kPa and 360 K. The reaction of barium hydroxide with ammonium thiocyanate is spontaneous but highly endothermic, so water, one product of the reaction, quickly freezes into slush. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
